A review of research on the efficacy of Viviscal, published in the September 2015 issue of the Journal of Drugs in Dermatology, suggests that the oral supplement may increase hair volume as well as the thickness of healthy, terminal hairs. ((Hornfeldt CS, et al. The Safety and Efficacy of a Sustainable Marine Extract for the Treatment of Thinning Hair: A Summary of New Clinical Research and Results from a Panel Discussion on the Problem of Thinning Hair and Current Treatments. J Drugs Dermatol. 2015 Sep;14(9):s15-22.)) The article presented more than two decades of research on the hair regrowth product and also included a discussion with a roundtable of dermatology and plastic surgery experts.
Both the research review and roundtable discussion point to the benefits of Viviscal, however the article’s conclusions can be questioned due to the appearance of a conflict of interest between the researchers and Lifes2good, Inc., the company that produces Viviscal. Additional independent research needs to determine if Viviscal is a viable and effective hair loss treatment.
Background
Viviscal, was launched in the U.S. in 2008 by Lifes2good Inc., Chicago, Illinois. Its key ingredient is a proprietary mix of powders derived from sustainably-harvested shark and mollusk species. The resulting “amino marine complex,” known as AminoMar C™, is blended with B and C vitamins, and minerals such as calcium to make Viviscal. The active ingredients in the AminoMar complex are called glycosaminoglycans (GAGs), a group of long chain sugar molecules present in many living creatures. GAGs hold water, and ingesting them may contribute to healthy hair and skin – although this is still speculative. According to Viviscal, the beneficial effect on skin and hair of a fish- and protein-heavy diet was first observed in Inuit people in the late 1980s. Viviscal is marketed primarily to women because of the relative dearth of effective hair loss treatments for female patients compared to men.
Review Article
The summary article by Hornfeldt, et al., ((Hornfeldt CS, et al. The Safety and Efficacy of a Sustainable Marine Extract for the Treatment of Thinning Hair: A Summary of New Clinical Research and Results from a Panel Discussion on the Problem of Thinning Hair and Current Treatments. J Drugs Dermatol. 2015 Sep;14(9):s15-22.)) notes that studies dating back to 1992 have suggested that Viviscal may treat hair loss to some degree. ((Lassus A, Eskelinen E, et al. A Comparative Study of a New Food Supplement, ViviScal®, with Fish Extract for the Treatment of Hereditary Androgenic Alopecia in Young Males. J Int Med Res. 1992 Nov;20(6):445-53.)) However, the more recent pivot to testing the supplement in women with thinning hair was pioneered by Dr. Glynis Ablon of the Ablon Skin Institute Research Center, Manhattan Beach, California. In a 2012 pilot study, Dr. Ablon found that Viviscal increased the number of terminal hairs by 211% and 225% after three months and six months, respectively. ((Ablon G. A Double-blind, Placebo-controlled Study Evaluating the Efficacy of an Oral Supplement in Women with Self-perceived Thinning Hair. J Clin Aesthet Dermatol. 2012 Nov;5(11):28-34.)) This was followed by a three month clinical study of women with self-perceived thinning hair; which the author attributed to poor diet, stress, hormones, or abnormal menstruation. In this study, published in early 2015, the mean number of terminal hairs increased by 32%, the count of shed hairs decreased by 39%, and subjects reported a significant increase in quality of life. ((Ablon G. A 3-Month, Randomized, Double-Blind, Placebo-Controlled Study Evaluating the Ability of an Extra-Strength Marine Protein Supplement to Promote Hair Growth and Decrease Shedding in Women with Self-Perceived Thinning Hair. Dermatol Res Pract. 2015; 841570.)) A similar randomized, placebo-controlled study, also led by Dr. Ablon and published in December 2015, found that female patients on Viviscal showed an almost 80% increase in terminal hairs and increase of 11.67% in hair diameter. ((Ablon G, Dayan S. A Randomized, Double-blind, Placebo-controlled, Multi-center, Extension Trial Evaluating the Efficacy of a New Oral Supplement in Women with Self-perceived Thinning Hair. J Clin Aesthet Dermatol. 2015 Dec;8(12):15-21.))
Some additional publications, such as Bloch’s 2014 study, ((Bloch L. Demonstrating the efficacy of a nutraceutical for promoting hair growth using a digital photography technique with posterior image analysis. Submitted for poster presentation at the 2015 World Hair Congress, Miami.)) suggest that Viviscal is effective in increasing patients’ hair volume and thickness. Another study published in 2014 suggests that Viviscal may improve scalp coverage and hair fullness in men with common baldness. ((Pinski KS. Patient satisfaction following the use of a hair fiber filler product to temporarily increase the thickness and fullness of thinning hair. Skinmed. 2014;12(5):278-281.))
In the roundtable discussion, which took place in August 2014, dermatology and plastic surgery physicians discussed findings of several clinical studies and reported a positive inclination to offer Viviscal as a treatment option. ((Hornfeldt CS, et al. The Safety and Efficacy of a Sustainable Marine Extract for the Treatment of Thinning Hair: A Summary of New Clinical Research and Results from a Panel Discussion on the Problem of Thinning Hair and Current Treatments. J Drugs Dermatol. 2015 Sep;14(9):s15-22.))
Limitations of Hornfeldt Review
The main limitation of the Hornfeldt article lies in the potential conflict of interest with the researchers and Lifes2good. Dr. Carl S. Hornfeldt received honoraria fees as a consultant for Lifes2good and his co-author of the review article, Mark Holland, is an employee of Lifes2good. Members of the expert roundtable advised Lifes2good on Viviscal or received an honorarium for their participation.
Summary
Viviscal has the potential to offer a new avenue of treatment for treating hair loss or supplementing current therapies. The review article provides a review of research and presents compelling findings over a span of two decades. However, more research is necessary into the long-term efficacy of Viviscal and the effects of glycosaminoglycans on the hair growth cycle. Also, given the appearance of a conflict of interest between the researchers and Lifes2good, it is particularly important that further research be conducted by independent investigators.
Posted by Robert M. Bernstein M.D. 


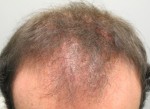 LPP in Male Pattern Alopecia
LPP in Male Pattern Alopecia A double-blind scientific study published in the May 2012 issue of the Journal of the American Academy of Dermatology has found that latanoprost, a drug that mimics naturally-derived compound molecules called prostaglandins, significantly increases hair density on the scalp after 24 weeks of treatment in young
A double-blind scientific study published in the May 2012 issue of the Journal of the American Academy of Dermatology has found that latanoprost, a drug that mimics naturally-derived compound molecules called prostaglandins, significantly increases hair density on the scalp after 24 weeks of treatment in young 
 Dr. Angela Christiano, a colleague of Dr. Bernstein’s at Columbia University, has been studying the causes of alopecia areata and genetic hair loss for many years. She, in fact, suffers from the disease as well.
Dr. Angela Christiano, a colleague of Dr. Bernstein’s at Columbia University, has been studying the causes of alopecia areata and genetic hair loss for many years. She, in fact, suffers from the disease as well. CBS News’ The Early Show has picked up the “balding buzz” that first started to grow when the National Enquirer reported that New England Patriots star quarterback Tom Brady is seeking advice on how to treat his hair loss.
CBS News’ The Early Show has picked up the “balding buzz” that first started to grow when the National Enquirer reported that New England Patriots star quarterback Tom Brady is seeking advice on how to treat his hair loss.


