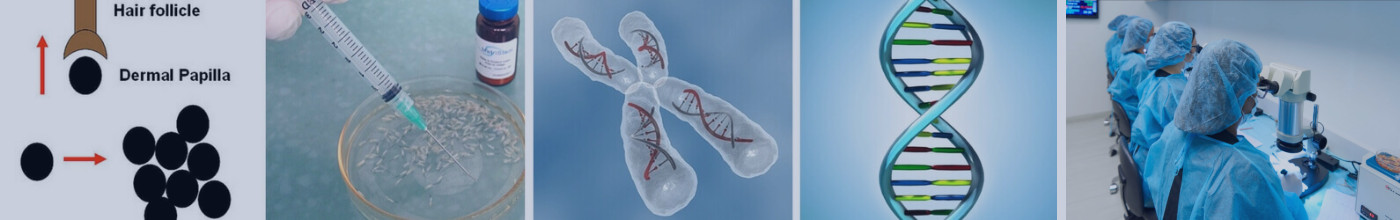
What is Hair Cloning?
Hair cloning and hair multiplication have been the subject of much research and discussion in the field of hair restoration. While these innovative techniques have yet to demonstrate consistent effectiveness in treating androgenetic alopecia, or common genetic hair loss, we remain optimistic about the potential for future breakthroughs. As a practice committed to staying at the forefront of hair restoration technology, we will continue to closely monitor any developments in this area.
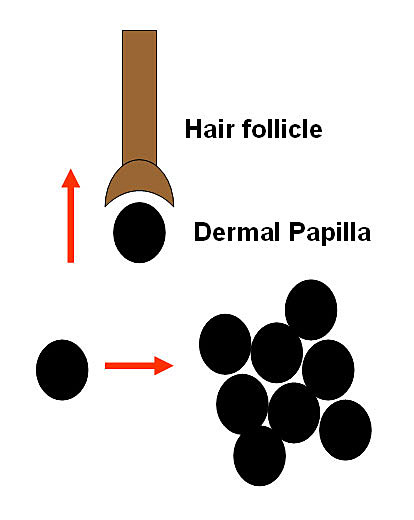
 Researchers are experimenting with ways to induce multiplied follicles to grow hair, including trials with ACell MatriStem
Researchers are experimenting with ways to induce multiplied follicles to grow hair, including trials with ACell MatriStemThe concept behind hair cloning involves multiplying a sample of a person’s germinative hair follicle cells outside the body (in vitro) and then re-implanting them into the scalp, with the goal of growing new hair follicles and, consequently, new permanent hair. Hair multiplication, on the other hand, focuses on removing donor hair follicles from the scalp and manipulating them to increase the total amount of hair, either by using transected follicles or plucked hair fragments.
Researchers have been exploring various methods to make these techniques viable, such as implanting dermal papillae cells, placing DP cells alongside miniaturized follicles, implanting DP cells with keratinocytes, and cell implantation using a matrix. While some progress has been made, there are still significant challenges to overcome, including determining the most suitable follicular components, successfully culturing cells outside the body, maintaining proper cell alignment, and ensuring consistent and natural-looking hair growth.
Safety is also a paramount concern, as any potential treatment must undergo rigorous testing and FDA approval to ensure that it does not pose risks, such as inducing tumors or exhibiting malignant growth.
Despite the challenges, we remain dedicated to exploring the potential of hair cloning and multiplication. Our team, including skilled providers like Dr. Bernstein, will stay abreast of any advancements in this field and evaluate their potential for incorporation into our practice.
While we await further developments, we continue to offer a range of effective, proven hair restoration treatments to our patients. Our experienced providers work closely with each individual to develop personalized treatment plans that address their unique needs and goals.
If you are experiencing hair loss and would like to learn more about your options, we encourage you to schedule a consultation with one of our knowledgeable providers. Together, we can explore the best course of action to help you achieve your desired results and restore your confidence in your appearance.
Rest assured that as hair cloning, hair multiplication, and other innovative techniques progress, we will be among the first to evaluate their safety and effectiveness, always keeping our patients’ well-being and satisfaction at the forefront of our practice.