Robert M. Bernstein, M.D., New York, NY and William R. Rassman, M.D., Los Angeles, California
Hair Transplant Forum International 2002; 12(1): 9-10.
It is surprising to find that patients with tight scalps and snug donor closures often heal with fine scars, whereas patients with loose scalps and easy to approximate wound edges occasionally heal with unacceptably wide scars. This seems contrary to the basic surgical dictum that non-tension closures heal better than those that are tight.
After seeing a patient with Ehlers-Danlos Syndrome several years ago, we began to think that connective tissue integrity was possibly more important than scalp laxity per se, in determining whether or not a primary closure would heal with a fine scar. It would help to explain the apparent “Scalp Laxity Paradox” – the sometimes-inverse relationship seen between scalp laxity and donor scarring (i.e. the better the laxity, the worse the scarring).
Case Study
A 26 year-old white male with male pattern alopecia presented to our office after having 6 hair transplant procedures between 1995 and 1999. Other than an unnatural, pluggy-looking frontal hairline, his first 4 procedures were uneventful. His 5th and 6th procedures healed with slightly widened donor scars. Our goal was to remove some of the larger grafts and re-distribute them as individual follicular units, in order to soften the appearance of his frontal hairline. In addition, we planned to excise the widest scar hoping to reduce its size and, in the process, harvest a small amount of hair to transplant to the frontal scalp. Since only some of the scars were wide, and the scalp was still lax, our clinical impression was that the widened donor scars were most likely technique dependent. The patient had no other abnormal scars on his body and he had a negative skin pull for Ehlers-Danlos Syndrome. Although we weren’t considering the diagnosis of EDS at the time, we perform this test routinely on all patients presenting with widened donor scars.
We harvested a 12.5 x 0.7 cm donor strip that yielded 235 follicular units from the periphery of the excised scar. These grafts were placed at the frontal hairline and in the anterior scalp. We closed the donor wound, without tension, using a 4-0 Monocryl running stitch. The procedure was uneventful. Post-operatively the patient developed mild but persistent erythema and edema along the suture line. There was no response to oral antibiotics. At 8 weeks post-op, with the symptoms persisting, our clinical impression was that the patient was possibly experiencing a hypersensitivity reaction to the Monocryl sutures (although the incidence of this is extremely low). We treated the area with a small amount of intralesional triamcinolone acetonide 10mg/cc injected along the suture line. At 10 weeks post-op, the scar had returned to its original width and we entertained a diagnosis of Ehlers-Danlos Syndrome (Figure 1).
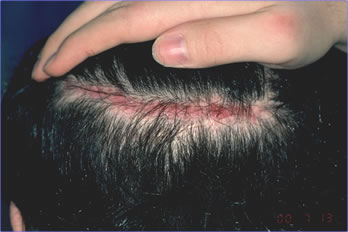 Figure 1. Widened donor scar at time diagnosis of EDS was considered.
Figure 1. Widened donor scar at time diagnosis of EDS was considered.The patient’s history was taken in greater detail. It revealed a number of symptoms that were not indicated by the patient in the history questionnaire or picked up by the doctor at the initial consultation. These included: 1) slow healing from testicular surgery in childhood, 2) back pain from kyphosis, 3) mitral valve prolapse, 4) chronic periodontal disease, and 5) undiagnosed chronic arthritis. On re-examination the patient was noted to have hyper-extensible joints and was able to touch his nose with his tongue (Figures 2 & 3). The patient was sent to the Department of Genetics at Schneider Children’s Hospital for further evaluation. Based upon his history and clinical findings, he was felt to have a diagnosis most consistent with Ehlers-Danlos Syndrome: Benign Hypermobile (Type III). There are no specific biochemical tests available for this type of EDS. Skin biopsies were taken from the patient for biochemical testing of cultured fibroblasts. Type I & III pro-collagen and collagen were examined by protein gel electrophoresis to rule out the more severe forms of EDS. These tests were normal.
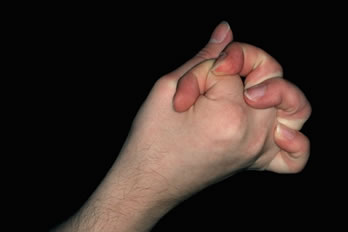 Figure 2. Joint hyper-mobility characteristic of EDS.
Figure 2. Joint hyper-mobility characteristic of EDS.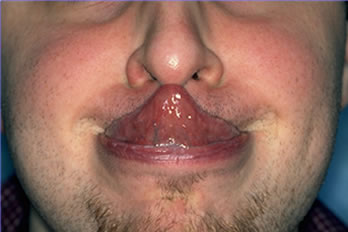 Figure 3. The ability to touch the tip of the nose with the tongue (Gorlin’s sign) is seen in 50% of patients with EDS.
Figure 3. The ability to touch the tip of the nose with the tongue (Gorlin’s sign) is seen in 50% of patients with EDS.Discussion
Ehlers-Danlos Syndrome is a group of inherited disorders of connective tissue characterized by one or more abnormalities of joint hyper-mobility, skin hyper-extensibility, poor wound healing, abnormal scarring and easy bruising. There are 11 clinical variants, or subtypes, that arise from a variety of abnormalities of collagen structure, function, synthesis, and/or catabolism. Six subtypes have known biochemical abnormalities of collagen. The incidence of EDS in the general population is 1:440,000 with approximately 12% having EDS Type III. ((Pinnell SR McKusick VA. Heritable Disorders of Connective Tissue with Skin Changes. In: Fitzpatrick et al., eds. Dermatology in General Medicine, 3rd ed. New York: McGraw-Hill, 1987.)), ((Demis DJ. Ehlers Danlos Syndrome. In: Clinical Dermatology, 21st Revision. 1994; (1) 4-3.))
Although our patient developed a widened donor scar, it was interesting that he did not have the classic “cigarette paper” wrinkled scarring seen in many cases of EDS, nor was he positive for the skin distensibility test (stretching the skin on the ventral forearm and measuring the elevation). We routinely use this extensibility test on all patients presenting with wide scars. The tests negativity possibly contributed to our not considering the diagnosis of EDS initially. The wide range of clinical symptoms of Ehlers-Danlos Syndrome raises the question of how many cases may actually go undiagnosed. One can certainly postulate that a forme fruste of EDS may be the cause of some of the unexplained cases of wide donor scars seen in surgical practices and may be a partial explanation for the Scalp Laxity Paradox seen in hair transplantation. It may also help to explain the “mush dermis” condition described by Dr. Dow Stough and why direct follicular unit extraction from the donor area is not possible in every patient.3
Dr. Gerard Seery’s excellent commentary in the Forum ((Seery G. Commentary #1. Hair Transplant Forum International 2001; 11(6): 179-180.)) on Cary Feldman’s article on “Tissue Laxity,” ((Feldman CS. Tissue Laxity based on Donor Tissue Ballooning. Hair Transplant Forum International 2001; 11(4): 119.)) gives additional insight into the Scalp Laxity Paradox. Dr. Seery describes two distinct contributors to scalp laxity: Glidability – the ability of the scalp to slide or glide over the underlying pericranium (related to the fibroareaolar layer of the scalp) and Extensibility – the ability of the scalp to stretch (related to the elastin content of the dermis). He states that these two factors, Glidability and Extensibility, are independent phenomena. Dr. Seery concludes that: “Some scalps are highly elasticized and reasonably wide strips can be removed purely by undermining and stretching, but this is relatively much more detrimental to tissue viability than sliding.”
There is another implication of this differentiation that can be of great importance to hair restoration surgeons. When scalp laxity is due to Glidability, one can be confident that a loose scalp will result in a fine donor wound. However, if scalp laxity is due to Extensibility, then “Surgeon Beware.” An extensible scalp may give the false impression that an easily closing wound will heal with a fine scar. Instead, it may be a signal that there might be excessive post-operative stretching and a cosmetically unacceptable result. In addition, the extensible scalp may be a sign of underlying connective tissue defects—or possibly EDS. If only we could differentiate between the two before the hair restoration surgery begins!
Dr. Feldman devised a means to determine “scalp elasticity” by injecting saline into the subcutaneous space and then assessing how much the tissue “balloons” as a result.5 Dr. Feldman implies by the term “scalp elasticity,” that he is actually measuring Extensibility as defined by Dr. Seery (i.e. the ability of the scalp to stretch due to the elastin content of the dermis). Dr. Seery describes a simple way to measure Glidability. “This is easily determined by simply placing the pulps of the examining fingers on the scalp and moving it on the underlying pericranium.”
So there you have it: we now have easy ways of measuring the two components of scalp laxity—well, not so fast! How do we know that Dr. Feldman’s test is not really measuring Glidability and Dr. Seery’s is not actually measuring Extensibility? Or that both are measuring a combination of the two; by assessing tissue laxities, but not differentiating which is the responsible component? I don’t think that we can really tell from these tests! How can we tell, for example, that the tissue “distension” measured by the balloon is due to skin stretch rather than from movement in the subcutaneous space or that when the skin is “moved” with the finger tips, it is simply gliding over the pericranium and not stretching a little into its new position?
Distinguishing between Glidability and Extensibility may be of more than academic importance. In clinical practice, the contribution of each may not be so easy to ascertain, but an accurate differentiation between these two causes of laxity may allow the surgeon to determine which patients may truly be at risk to form wide donor scars. It is possible that biochemical evaluations on patients with loose scalps may uncover a spectrum of conditions characterized by borderline defects in connective integrity and may serve an adjuvant to the clinician managing patients with wide scars. A deeper knowledge of the structure and function of connective tissue in patients without overt clinical syndromes may be the real key to understanding the Scalp Laxity Paradox.






