Robert M. Bernstein, MD, New York, NY
William R. Rassman, MD, Los Angeles, CA
Dermatologic Surgery 1998; 24(8): 875-880.
Robert M. Bernstein, M.D. Assistant Clinical Professor of Dermatology, College of Physicians and Surgeons, Columbia University, New York, NY William R. Rassman, M.D.
With the increasing popularity of “follicular unit transplantation,” ((Bernstein R, Rassman W, Szaniawski W, Halperin A: Follicular Transplantation. International Journal of Aesthetic and Restorative Surgery 3:119-132, 1995.)), ((Bernstein RM, Rassman WR: Follicular Transplantation: Patient Evaluation and Surgical Planning. Dermatologic Surgery 23:771-784, 1997.)), ((Bernstein RM, Rassman WR: The Aesthetics of Follicular Transplantation. Dermatologic Surgery 23:785-799, 1997.)) in which naturally occurring, individual follicular units are used exclusively in the hair restoration procedure, and with the generally increased awareness that the integrity of these units must be preserved regardless of the method of transplantation, the need to determine the optimal way to dissect intact follicular groups from the donor strip has become extremely important.
Loop magnification with back lighting (transillumination), originally developed by Dr. Paul Rose in 1995 (Rose P: Presentation, ISHRS Meeting, Nashville, Tennessee, Sept. 1996), aids in the visualization and dissection of follicular units and has been especially useful in patients with blond or white hair. The use of the microscope to facilitate graft dissection was first advocated by Dr. Bobby Limmer who has been using this technique since 1988. ((Limmer BL: Elliptical donor stereoscopically assisted micrografting as an approach to further refinement in hair transplantation. J Dermatol Surg Oncol 20:789-793, 1994.)) He has long held that the microscope offers significant advantages over direct visualization and/or loop magnification. Dr. David Seager has observed 20% more hair when using the microscope ((Seager D: Binocular stereoscopic dissecting microscopes: should we all be using them? Hair Transplant Forum International Vol. 6, No 4:2-5, 1996)), but this has not been documented in a well controlled study. Dr. Norwood had also felt that the microscope produced a better yield, but cautioned that switching to the microscope could throw an office into “turmoil” if the transition is made too abruptly ((Norwood OT: Notes from the editor emeritus: Limmerization. Hair Transplant Forum International Vol. 6 No 6:12, 1996)). In the experience of these authors, the microscope was well received and quickly adapted by our staff, although in the initial training phases, follicular dissection took somewhat longer.
Other practitioners have also expressed concerns that the use of the microscope may unduly slow down the procedure and that staff resistance to this new technology may be an insurmountable problem in some practices. Dr. Richard Shiell has questioned the “20% wastage factor,” and has suggested that experienced technicians can cut excellent grafts without the microscope ((Shiell R: Limmerization; Editor’s comments. Hair Transplant Forum International Vol. 6 No 6:13, 1996)). He also questioned whether the higher economic costs due to the purchase of microscopes, staff training, and slower dissection, would outweigh the benefits of using a “state of the art microscopic technique.” Another issue addressed by Dr. Shiell is the question of whether an increase in yield on the order of 10 to 20% is even critical to the transplant process since, in his opinion, less than 20% of men (and only 2% of the hair transplant population) loose so much hair that their donor supply would be a problem later in life. Dr. Norwood, on the other hand, feels that 95% of hair transplant patients would eventually need to use all available donor hair (Personal communication with Dr. O’Tar Norwood, October 1996) so that obtaining the maximum yield from the donor source is very important.
It is the opinion of these authors that the limitations of the donor supply are critical to the outcome of the transplant in those patients whose donor supply may be maximally exploited. Therefore, the donor supply must be accurately assessed and carefully considered in each and every patient undergoing surgical hair restoration.1,2 In addition, since relatively large numbers of follicular units must be utilized if the patient is to achieve satisfaction in a limited number of transplant sessions,2 we feel that even relatively small increases in either the number or quality of follicular units would be of significant benefit to the patient, and would justify using more accurate dissecting techniques.
Dr. Seager has shown that breaking up follicular units into individual hairs follicles can result in poor graft growth.5 Drs. Cooley and Vogel have shown that delayed graft growth may be caused by the removal of the dermal papillae during dissection ((Cooley J, Vogel J: Loss of the dermal papillae during graft dissection and placement: another cause of x-factor? Hair Transplant Forum International Vol. 7 No 1:20-1, 1997)). Both of these studies indicate that meticulous graft dissection may not only increase the absolute number of implants obtained from the donor strip, but can improve their growth as well, thus lending further support to the importance of precise dissecting techniques.
The purpose of this study was to objectively measure the yield of follicular implants obtained with the dissecting microscope as compared to using loop magnification with transillumination (back-lighting). This study does not attempt to address the question of how the two dissection techniques will affect implant survival. This important question still needs to be addressed in well controlled, prospective studies.
Our facility was in a unique position to examine the relative merits of the different methods of follicular dissection. We have been performing follicular unit transplantation since the beginning of 1995,1 and had developed significant expertise in dissection both with, and without transillumination. Dissection with transillumination and loop magnification had been fully implemented in our clinic since December 1996. At the time we implemented transillumination with loop magnification, we performed a cursory study of graft yield and felt that the benefits of this technique over conventional “tongue blade” dissection were on the order of 20%. Prior to fully converting to the microscopic, we were determined to objectively ascertain the value gained by switching to this type of dissection.
At the time this study was conducted we were performing part of each procedure with the microscope. Our decision to fully adopt microscopic dissection would be based upon the benefits of increased implant yield, weighted against greater dissection time and staffing issues.
Although our data was accumulated after the quality of our dissection appeared to be stable, it is important to emphasize that the study was conducted in a transitional period, during which time our staff had great facility in one technique (magnification dissection with back-lighting) and more limited experience with another (microscopic dissection). Discussions with medical groups who have had longer experience in microscopic dissection, suggest that further benefits with the microscope may be obtained over time, and that the learning curve when using this technique does not plateau quickly. The results of the present study should be viewed in this context. This study is, therefore, not meant to be a definitive work on the merits of the microscope, but rather to examine quantitatively, and in a more controlled way, some of the impressions that have, thus far, been arrived at anecdotally.
Materials and Methods
This study was prospective, randomized, and bilaterally controlled. All patients in the study were undergoing their first elective hair restoration procedure for male pattern baldness. Follicular unit transplantation1 was used exclusively in the protocol.
Local anesthesia consisting of a mixture of lidocaine, bupivicaine, and epinephrine was administered in a ring block fashion. A strip of donor hair was clipped to approximately 2-3 mm in length. The donor strip was harvested from the mid-portion of the permanent zone in the back of the scalp. A two-bladed knife was used to standardize the width of the donor strip. The strips ranged from 1.2 to 1.8 cm in width and from 12 to 30 cm in length. Dissection was carried out in the mid to deep fat, below the level of the hair follicles. The wound was closed with a single, running, cutaneous suture.
The entire donor strip was dissected and transplanted, but only the medial aspect was used in the study in order to minimize variations in the density of each side as one moved laterally. The harvested strip was bisected at the midline using a straight razor. The two pieces were then placed side-by-side so that the midline edges aligned flush with one another. The lateral aspects of each strip were removed so that the medial portions to be studied were of equal length. The size of the medial portions of the strip devoted to the study varied from procedure to procedure, but was always equal.
The two strips were randomly assigned for dissection using the microscope (group A) or using loop magnification with transillumination (group B). Each piece was further subdivided into approximately 3 mm vertical sections. This was accomplished with a straight razor making a single vertical cut through the strip, under magnification, while traction was applied to each end of the strip, with the help of a second assistant. During the course of the dissection the technicians performing the study would alternate between using the microscope and using the loops. In this way, fatigue would have a minimal effect on the outcome. Our most experienced technicians were used for the study and were chosen after they had reached a steady level of competence using the microscope. Other technicians assisted in dissecting the lateral aspects of the strip not used in the study.
The individual strips were laid on their side and dissection was accomplished using a #10 Persona blade, set on a number 3 blade handle. The staff was instructed to make every attempt to keep the follicular units intact. Excess skin between the follicular units was trimmed away. As they were dissected, the implants were sorted into groups containing 1, 2, 3, and 4 hairs. All hair fragments that were judged to be potentially viable were counted and used in the study. This judgment was carefully made under physician supervision and was the same for both groups. The waste was also checked for potentially viable hair. Implants of both groups were spot checked under the microscope for a subjective evaluation of the quality of the dissection. The time spent for each type of dissection was also recorded.
Microscopic dissection was performed using a Meiji-ETM Microscope at 10x power. Standard magnifying loops ranging from 1¾ to 2½ magnification were used. The choice of specific loop magnification depended upon the personal preferences of the dissectors, with 2x being the most common power. Custom made back-lighting tables, based upon the design of Dr. Paul Rose, were used for transillumination.
Results
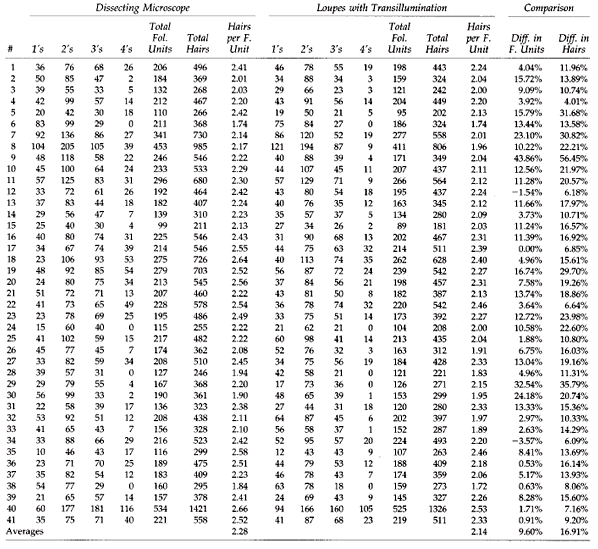
Table 1 describes the number and composition of follicular units generated using the dissecting microscope and loop magnification with transillumination in the 41 patients studied. Microscopic dissection produced, on the average, 10% more follicular units than loop dissection. The average number of hairs per implant was 2.28 with the microscope, and 2.14 with loop dissection. The average total number of hairs generated by the microscope was 17% greater than with the loops.
Although not the focus of our study, a subjective spot microscopic evaluation of the quality of the implants from each group revealed that the microscopically dissected implants were generally of the same size as the loop dissected group. The microscopically dissected grafts were sometimes trimmed more evenly, but in general an independent observer could not distinguish between the implants dissected by using the microscope from those dissected with the loops. We did note a small number of hair bulbs left exposed (not covered by subcutaneous fat) in the loop group, but this was also noted in the microscope group. It was most commonly seen involving one hair of a larger follicular unit. In some cases, it may have represented uneven harvesting, rather than being related to the dissection per se. A more rigorous evaluation of implant quality has been carried out by Drs. Jerry Cooley and James Vogel (Cooley J, Vogel J: Follicle trauma in hair transplantation: Prevalence and prevention. Presentation, ISHRS Meeting Barcelona, Spain, Oct. 1997).
As expected from the numerical differences in hair counts, there were less hair fragments in the discarded tissue resulting from microscopic dissection than from loop dissection. The discarded tissue from both methods of dissection was always scrutinized for possible viable follicles, and questionable fragments were always counted and planted.
The average time for dissection with the microscope was approximately twice as long as with the loops when taken at the point in which the technician first reached a level of competence high enough to generate quality implants. With experience, the technicians time for microscopic dissection continued to decrease but seemed to reach a stable level when it took approximately 25% longer than with loop dissection.
Table 1. A comparison of follicular unit and hair counts using different dissecting techniques.
Discussion
The results of our study clearly show an increase in the yield of follicular unit grafts (implants) from the donor strip when using the dissecting microscope, and supports the increase in yield observed by others.5 The results are even more significant when the total numbers of hairs are measured. The reason is that microscopic dissection not only yielded more follicular unit implants, but each implant contained, on the average, slightly more hair. It is important to emphasize that the 17% gain in the total number of hairs obtained with the microscope was an increase over that achieved with loop magnification and transillumination. If microscopic dissection was compared to unaided direct visualization, the differences would be even more dramatic.
In our protocol, we made vertical subdivisions in the strip at 3mm intervals, under direct visualization with the loops, but prior to microscopic dissection. This enabled our technicians to work with smaller pieces of tissue and to immediately lay the strip on its side. From this position, all the microscopic dissection could easily be performed on small strips that were well stabilized on the cutting surface.
The method originally devised by Dr. Limmer (Personal communications with Dr. Bobby Limmer, November 1997), and adapted by others (Personal communications with Dr. David Seager, October 1997), is to initially subdivide the strip, under microscopic control (to completely avoid transecting follicular units) into several large sections. This initial sectioning, performed by the most proficient staff member, generates tissue for the other experienced staff members to work with. These staff members, beginning at one end of their large section, generate slivers that are one follicular unit deep. These fine slivers are then laid on their sides and the dissection is easily completed by the rest of the staff.
The current technique used by Dr. Limmer (Personal communications with Dr. Bobby Limmer, November 1997), is designed to more evenly distribute the workload to the staff. In this method, the initial strip is vertically subdivided into approximately 2.0 – 2.5 mm wide pieces under microscopic control. The other staff members, starting at one end of each piece, complete the dissection under the microscope. We adopted this technique in our facility once the study had been completed. Although it is initially more difficult and slower, the yield is greater with complete microscopic dissection, possibly adding an additional 5% or more, to the 17% benefit already achieved with using the microscope for only the dissection of the individual pieces. In retrospect, we feel that the method offered in our protocol might be appropriate for a “team in transition,” but with more experience the microscope should be used to guide the initial vertical sectioning as well. It is obvious that complete microscope control of every aspect of the dissection should be the goal.
It has been our subjective impression that less skilled cutters may actually show more improvement in their dissection from using the microscope than those that are highly trained. If this is the case, dissecting teams with more limited experience may have even greater benefit from using the microscope. Dr. Seager also believes that the value of the microscope may be more significant in the hands of the less experienced dissectors. As we alluded to in the introduction, some continued improvement may possibly be achieved when the dissectors use the microscope over extended periods. Dr. Seager feels that there is at least an additional 5% increase in yield during the second 6 months. Interestingly, he has found that with continued practice, the microscope may take only 10% longer, but can take 4x times as long in the first month (Personal communications with Dr. David Seager, October 1997).
Another advantage of the microscope, besides the increased yield and the easy identification of follicular units, may be decreased back and neck strain. This is probably due to the more upright posture of the staff when using the stereoscope compared to the loops.
The main disadvantage of using the microscope has been the increased dissection time. Ergonomically, there is inefficient movement of grafts in and out of the microscope’s visual field. Better organization of the dissected implants at the periphery of the field, appears to reduce the need to constantly refocus one’s eyes and therefore speeds up the dissection process. In addition, not trimming the implants too close will decrease dissection time, and most likely maximize graft survival. As we have shown, the dissecting time decreases significantly as the staff becomes more accustomed to using the stereoscope.
In the office where this study was performed, the microscope was presented as a new tool to possibly improve the capabilities of the staff, and learning microscopic dissection became an absolute job requirement. In this setting, the new procedure and was met with much interest and enthusiasm.
The main advantages of transillumination are that: 1) It does not represent a significant break from traditional dissecting techniques, so that the staff more readily adapts to this change. 2) Portable back-lighting surfaces are relatively inexpensive, and are smaller, less fragile, and easier to store than dissecting microscopes.
There are a number of problems that we have encountered with transillumination: 1) Built-in back-lighting stations limit the usefulness of that surface for other purposes. 2) To be maximally effective, the overhead lights need to be dimmed. This hampers surgical activities performed in the room at the same time, such as creating recipient sites. 3) As the cutting surface becomes scratched, the light source becomes partially blocked, limiting the ability to perform accurate dissection. 4) Unless the cutting surfaces are disposable, they are very difficult to clean and sterilize. 5) Most importantly, the usefulness of back-lighting is severely limited in all but the thinnest of sections, and is virtually useless in the initial subdivision of the donor strip. In other words, in order for back-lighting to be effective, the strip must first be prepared by dissecting techniques which cut the strip under low visibility, and risk the integrity of the follicular unit.
Although not examined in this study, we have found that the microscope offers additional advantages in cases where there is donor scarring and concomitant distortion of the hair follicles. Similarly, we have found that the microscope is especially useful in dissecting the curved hair shafts characteristic of the black races.
A criticism of extensive micrografting has been the resultant thin look. It is clear that in order to truly benefit from the follicular unit transplantation procedure, the full follicular unit must be preserved in the dissection. The use of the multi-bladed knife with more than two blades (which had been used by our group in the past and has since been abandoned), also does not permit the surgeon to capture intact follicular units as they are broken up even before the dissection takes place. When this is combined with less than optimal dissecting techniques, the results approach that of micrografting. If, because of poor harvesting techniques and less than optimal graft dissection, only partial follicular units are transplanted, the surgeon cannot possibly expect to achieve the best aesthetic results.
The fullness achieved by using significant numbers of the naturally occurring three and four hair implants obtained from careful follicular dissection, produces a cosmetic impact that cannot be duplicated with the one and two hair grafts that result when the integrity of the larger follicular groups are not preserved in the dissection. However, attempts to combine multiple follicular units to create greater density will never match the naturalness produced by using the intact follicular unit, since unnecessary skin is moved and larger wounds must accommodate the larger skin volume.1,2 The goal, therefore, should be to preserve the follicular unit in the harvesting and in the dissection process. Once the naturally occurring follicular units of various sizes are identified and dissected, their selective distribution can be used to produce the best possible cosmetic results for the patient.3
Although recent evidence has suggested, and logic dictates, that preserving the tissue surrounding each follicular unit enhances graft survival, the question of whether the patient is better served when the follicular implants are carefully sculpted or merely trimmed of excess tissue still needs to be determined. The major advantages of close trimming the grafts are that it allows for smaller wounds, closer placement of recipient sites, and a resultant increased density, provided, of course, that the follicular unit is left intact. Very close trimming, however, may increase the chance of graft injury both during dissection and with subsequent graft handling and placement. Certainly, close trimming of the follicular units can only be accomplished safely by using the microscope.
Each practitioner must make judgments regarding how close to trim the implants based upon the skill of the staff and the goals of the surgery. The staff can be trained to use the microscope in a more expedient manner by trimming the follicular units less closely. Although this may increase cutting speed, it will not take full advantage of the capabilities and benefits of microscopic dissection. In the end, judgments regarding graft dissection become as much an art as other aspects of the transplant procedure, but no less critical to its outcome.
Conclusion
The growing popularity of the stereo-microscope can be justified by the increased yield that it achieves, especially in the patient who will fully exploit his, or her, donor supply. Greater absolute numbers of follicular units and increased hair counts in the individual grafts have been observed and documented with microscopic dissection when compared to loop magnification with back-lighting. Although microscopic dissection represents a significant departure from the “old school” of graft dissection, the new requirements of follicular unit transplantation, and other transplant procedures which require the increasing use of very small grafts, mandate more sophisticated dissecting techniques.
Some of the barriers to microscopic dissection can be lessened by having a transitional period in which a limited number of staff members use the microscope for only a part of the dissection, while the team gradually builds up confidence, technical competence, and speed. The technique of subdividing the donor strip into smaller pieces may also aid in this transitional process. Even for a “team in transition” the use of the microscope offers many tangible benefits over other methods of graft dissection. These advantages will be even greater when complete stereo-microscopic dissection is used.
The limiting factor in all hair restoration procedures is the patient’s finite donor supply. Meticulous stereo-microscopic dissection can help us preserve this supply, and ultimately provide the patient with more transplantable hair. As surgeons, we should make every effort to avail our patients of any technology that will improve the procedures that we perform.
Acknowledgment
The authors would like to thank Rebecca Sipala for her assistance in the preparation of this manuscript.





