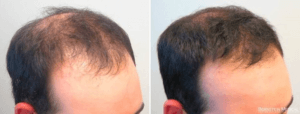
A 23-year-old male presents with a 3-year history of progressive thinning at his frontal hairline and crown. He has not tried any hair loss treatments. His father and maternal grandfather are nearly bald with only a small fringe of hair remaining at the back and sides of the scalp. He is fearful that he will slowly begin to look like them. He has no medical problems and does not take any medications. He reports that his father had prostate cancer.
Q: What would be the initial step?
- Start finasteride
- Start minoxidil initially and later considering adding finasteride
- Scalp biopsy
- Trichoscopy
Answer D. Before beginning medication, trichoscopy needs to be performed to verify the diagnosis. While the history and presentation are suggestive of androgenetic alopecia, trichoscopy should be done initially to verify that the hair has miniaturized which is, in addition to the characteristic pattern, the hallmark of androgenetic alopecia.
Q: What would be the best therapy to begin with?
- Minoxidil
- Minoxidil and consider finasteride in the future if minoxidil does not provide a good result
- Minoxidil and Finasteride
- PRP
Answer C. The primary cause of genetic pattern thinning is follicular sensitivity to dihydrotestosterone (DHT). Men with AGA have a high genetic sensitivity on the front and top of the scalp to DHT which causes the hair in these areas to become progressively miniaturized over each successive growth cycle. The most effective treatment is by blocking the DHT effects with finasteride. Minoxidil works by a different mechanism i.e., by increasing the length of the anagen cycle and its effects are synergistic to finasteride.
Q: The patient reports that his father had prostate cancer and heard that finasteride causes prostate cancer or can make prostate cancer worse, so he should not take it. What is the correct response?
- Finasteride increases the risk for prostate cancer, so patients with a family history should not be given the prescription.
- Finasteride can make prostate cancer worse, so patients with a family history of prostate cancer should not take finasteride. The reason is that finasteride shrinks normal prostate tissue and allows the cancerous tissue to grow unchecked.
- Finasteride increases the Gleason score of patients with prostate cancer and this is additional evidence that it makes prostate cancer worse.
- None of the above
Answer D. Finasteride at the 5mg concentration decreases the risk of prostate cancer (about 20%), but it is not FDA-approved for cancer prevention. Since finasteride shrinks hyperplastic (but non-cancerous) prostate tissue, the likelihood that a needle biopsy will hit the cancerous tissue is increased. This sampling error is felt to explain why the Gleason scores of the patients on finasteride were increased. Long-term follow up found no difference in morbidity between the finasteride and control groups. When explaining this to patients, it is important to stress that there is still some disagreement among urologists on this issue.
Q: The patient relates that he was told that finasteride can make the screening for prostate cancer more difficult. Which of the following is correct?
- The patient relates that he was told that finasteride can make the screening for prostate cancer more difficult. Which of the following is correct?
- Finasteride makes PSA a less sensitive screen for prostate cancer.
- Finasteride has no effect on prostate cancer screening.
Answer A. Since finasteride shrinks hyperplastic prostatic tissue, the 5mg dose generally lowers PSA by about 40%. It is not clear the degree to which the 1mg will decrease PSA. There is no evidence that finasteride makes the screening less sensitive. If there are concerns about prostate cancer, or the patient is at risk due to age or ethnicity, it is best to obtain a PSA before beginning finasteride and then repeat the test in a month to get a sense of the decrease on the drug. This level can then be used for further follow-ups. In these cases, it is important to have the patient’s urologist involved in the initial decision to start finasteride as well as the follow-up.
Q: The patient is concerned about taking oral pills and asks, “what is the main side effect of finasteride?” You answer:
- Testicular pain
- Brain fog/mood changes
- Sexual dysfunction
- Prostate cancer
- All the above
Answer C. The main side effect of oral finasteride is sexual dysfunction. It is seen in 2-4% of patients who use the medication. The main sexual changes observed are loss of morning erection, trouble obtaining or maintaining erections, or a decrease in sex drive. Testicular pain is present in less than 1% of men. Brain fog and depression if real, are rarely isolated symptoms (i.e., they generally occur in conjunction with sexual dysfunction).
Q: The patient has read about sexual dysfunction side effects and is wary because online forums report a syndrome called Post-Finasteride Syndrome (PFS). What is this syndrome?
- PFS is a syndrome of sexual dysfunction and mood disorders that are temporary.
- PFS is a situation where someone thinks they have side effects after they have been off finasteride.
- PFS is a syndrome that has been fabricated on the internet and is not actually real.
- Lowering the dose of finasteride can be helpful to prevent or alleviate PFS.
Answer B. Post-finasteride syndrome (PFS) is a constellation of serious adverse side effects of finasteride (whether real or imagined) that persist after discontinuing finasteride. The diagnosis is generally entertained after the patient has been off finasteride for three months.
Q: Are there any risk factors for developing post-finasteride syndrome?
- Older age when starting finasteride
- Using higher doses of finasteride than prescribed
- Psychiatric disease, such as OCD, anxiety, panic disorder, depression, body dysmorphic disorder and hypochondriasis
- All the above
Answer C. In our experience, the major risk factor for developing PFS is OCD with hypochondriasis and panic disorder second and third. The other psychiatric diseases also play a role. The literature suggests that the incidence of PFS increases with age, but in our experience, this is mainly a condition of young adults. The conditions appear to be idiosyncratic and not dose-related.
Q: The patient read that fertility is a concern. What do you instruct the patient to do regarding his finasteride when he is ready to start a family? (Select all that apply)
- He should not take finasteride when trying to conceive.
- He will need to temporarily stop finasteride when trying to have a baby and can resume after conception.
- He can take finasteride through conception but should stop it if he will be in close contact with a pregnant woman.
- He can continue the medication throughout conception and pregnancy.
- If the patient has difficulty conceiving (greater than 4-6 months), he should temporarily go off the medication.
- Pregnant women should not ingest the medication or handle broken pills.
Answer D, E, and F. Patients can remain on finasteride throughout conception and pregnancy. Infertility is listed as a potential concern with finasteride but is not normally an issue for most couples trying to conceive. Most couples conceive successfully while men continue taking finasteride. However, an exception would be if a couple has trouble conceiving after 4-6 months and a reproductive work-up for both genders fails to reveal the cause, then one should consider temporarily stopping finasteride. Men do need to be wary that they should wash their hands after handling finasteride. Women who are, or who may become pregnant should not have exposure to finasteride either orally or through handling crushed tablets due to possible teratogenic effects.
Q: The patient wants to start finasteride but wants to know what to expect. You should tell him which of the following? (Select all that apply)
- If there are no results in the first 6 months, then he should stop the medication because it is unlikely to work.
- Shedding is very common in the first few months of treatment.
- If there is significant shedding, the medication should be continued anyway.
- It usually takes about a year to see the full response to the drug.
- The medication works mainly in the crown.
- Finasteride is only indicated in preventing hair loss in younger patients. The medication can later be stopped, and hair transplant can replace the need for medication.
Answer C and D. In contrast to minoxidil, where significant shedding is common, the shedding with finasteride is generally mild. That said, if there is significant shedding the patient should be encouraged to continue the medication as it means it is working. Medical therapy works wherever there is some existing (miniaturized) hair, whether in the front of the scalp, the top, or the crown. It will not work in areas that are completely bald. Finasteride should be continued indefinitely to prevent further thinning (if there are no side effects). A hair transplant does not prevent the progression of hair loss and is thus not a replacement for medical therapy.
Q: What dose should one consider using during therapy?
- 1mg
- 1.25mg
- 2.5mg
- 5mg
- All the above
Answer E. The FDA-approved dose for hair loss is 1mg. It is best to always start with this dose as the package insert of the 1mg dose describes its use for androgenetic alopecia. The 5mg dose is FDA-approved for prostatic enlargement. After the patient had been on the 1mg dose, many doctors advise patients to take ¼ of the 5mg dose on their renewal. This dosing is less expensive and there appears to be some counterfeit 1mg pills (particularly when obtained through the internet, rather than from a physical drug store). After 3-5 years on the medication, many patients become relatively resistant to finasteride and a higher dose is required to maintain their hair. When you increase the dose in patients who have become relatively resistant, there does not seem to be an increased risk of side effects. That said, whenever using a dose other than 1mg, it is important to advise patients that this is an off-label dosing and that the side effect profile is not known.
If you are experiencing gradual hair thinning, please contact our office for an appointment. Call 212-826-2400 or schedule a consult here.





